Media Briefing/Press Conference
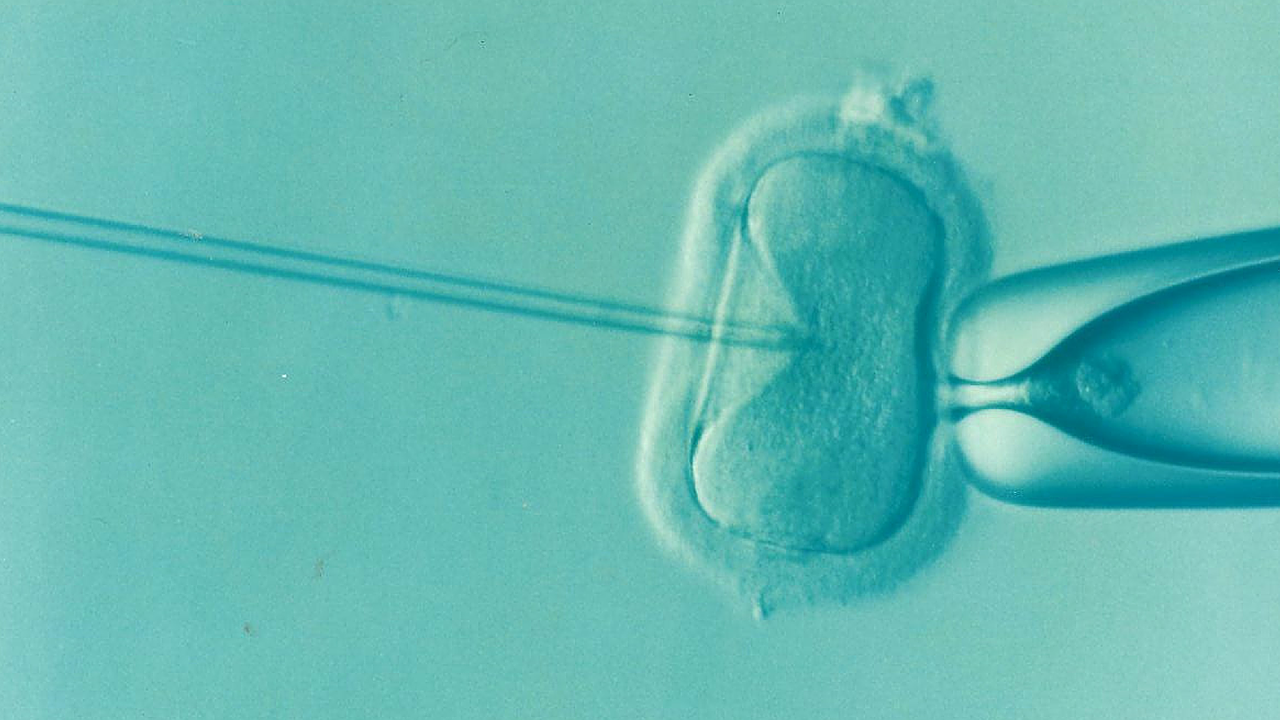
From: Australian Science Media CentreAround 1 in 25 women who give birth in Australia use some form of assisted reproductive technology (ART) to achieve their pregnancy. The use of ART, however, is not without controversy and there are important ethical considerations in both the clinical and research contexts.
The National Health and Medical Research Council (NHMRC), through the work of the Australian Health Ethics Committee (AHEC), has a well-established role in the provision of ethical advice for ART. On April 20 the CEO of NHMRC will issue the Ethical guidelines on the use of assisted reproductive technology in clinical practice and research, 2017 (the ART guidelines).
The previous ART guidelines were issued in 2004, with only the research guidelines updated in 2007. The 2017 update ensures that the ART guidelines provide contemporary advice to support the ethical conduct of ART in clinical practice.
Fertility and IVF clinics must adhere to these ART guidelines in order to be accredited.
This briefing will present the guidelines and will cover a number of issues including:
- information giving, counselling and consent requirements
- sex selection for non-medical purposes
- the use of donated gametes and embryos
- surrogacy
- pre-implantation genetic testing
- the collection and use of eggs and sperm from persons who are deceased or dying, and the use of stored eggs, sperm and embryos after the death of a person.
Copies of the embargoed material will be available from 17:00 AEST Wednesday 19 April on Scimex for journalists with a login. They are also available on request - contact us on (08) 7120 8666 or by email.
Further information about the development of the ART guidelines is available on the NHMRC website.
Speakers:
- Professor Anne Kelso AO is CEO of the National Health and Medical Research Council
- Professor Ian Olver AM is Chair of the Australian Health Ethics Committee, NHMRC
Date: Thu 20 April 2016
Start Time: 09:00am AEST
Duration: Approx 45 min
Venue: Online

Expert Reaction
These comments have been collated by the Science Media Centre to provide a variety of expert perspectives on this issue. Feel free to use these quotes in your stories. Views expressed are the personal opinions of the experts named. They do not represent the views of the SMC or any other organisation unless specifically stated.